Presurgical Applications of FMRI in Epilepsy
Funding source: NINDS R01 NS035929 (Binder, PI)
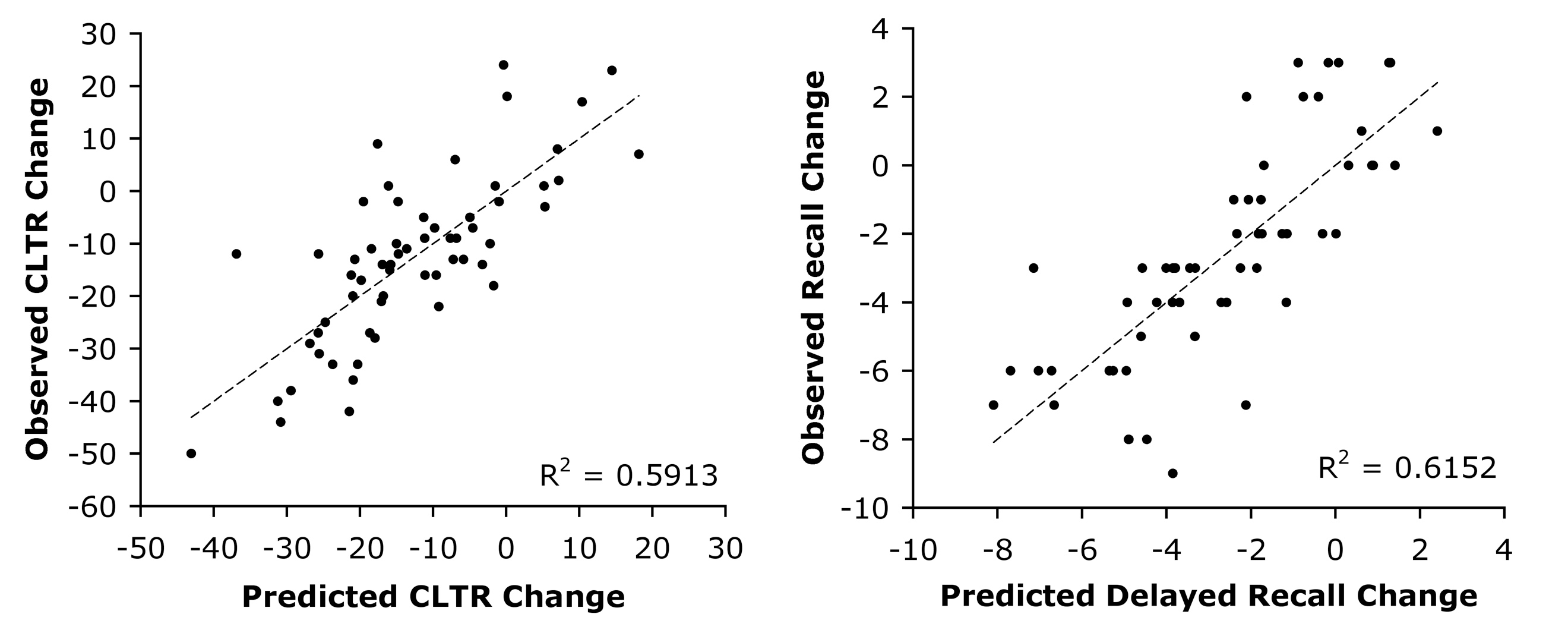
Surgical removal of the epileptic focus is an effective treatment for drug-resistant temporal lobe epilepsy (TLE), however patients who undergo surgery on the language-dominant (typically left) temporal lobe are at risk for developing new language and verbal episodic memory deficits. This project began in 1995 with the aim of developing and validating a non-invasive fMRI method for determining language lateralization as an alternative to the intracarotid anesthesia (Wada) test. Our database now includes over 250 prospectively enrolled volunteers with drug-resistant focal epilepsy who participated in standardized MRI and neuropsychological testing protocols before and after surgery. Our main focus has been on optimizing multivariate models for predicting naming and verbal memory outcomes after left anterior temporal lobe (ATL) resection. Published work to date has validated an fMRI protocol (Semantic Decision vs. Tone Decision) against the Wada test in a large sample of 229 patients. The fMRI language lateralization measure was shown to be superior to Wada results in predicting postoperative cognitive outcomes using regression models and in a sample of left TLE patients with discordant fMRI and Wada results.
In 2012, we formed the FMRI in Anterior Temporal Epilepsy Surgery (FATES) consortium with 9 other collaborating epilepsy centers (Cleveland Clinic, Emory University, Medical University of South Carolina, University of Alabama, UCLA, University of Cincinnati, University of Rochester, University of Washington, and Vanderbilt University) to accelerate enrollment of patients undergoing left ATL surgery and to validate our methods across centers and imaging platforms (including GE, Siemens, and Philips scanners). Analysis of the full dataset is underway.
Relevant Publications
- Binder JR, Sabsevitz DS, Swanson SJ, Hammeke TA, Raghavan M, Mueller W. Use of preoperative functional MRI to predict verbal memory decline after temporal lobe epilepsy surgery. Epilepsia, 2008; 49: 1377-1394.
- Binder JR, Swanson SJ, Hammeke TA, Sabsevitz DS. A comparison of five FMRI protocols for mapping speech comprehension systems. Epilepsia, 2008, 49: 1980-1997.
- Binder JR, Swanson SJ, Sabsevitz DS, Hammeke TA, Raghavan M, Mueller WM. A comparison of two fMRI methods for predicting verbal memory decline after left temporal lobectomy: Language lateralization vs. hippocampal activation asymmetry. Epilepsia, 2010, 51: 618-626.
- Binder JR, Gross WL, Allendorfer JB, Bonilha L, Chapin J, Edwards JC, Grabowski TJ, Langfitt JT, Loring DW, Lowe MJ, Koenig K, Morgan PS, Ojemann JG, Rorden C, Szaflarski JP, Tivarus ME, Weaver KE. Mapping anterior temporal lobe language areas with fMRI: A multi-center normative study. Neuroimage, 2011, 54: 1465-1475.
- Gross WL, Binder JR. Alternative thresholding methods for fMRI data optimized for surgical planning. Neuroimage, 2014, 84: 554-561.
- Janecek JK, Swanson SJ, Sabsevitz DS, Hammeke TA, Raghavan M, Rozman M, Binder JR. Language lateralization by fMRI and Wada testing in 229 epilepsy patients: Rates and predictors of discordance. Epilepsia, 2013, 54(2): 314-322 (PMCID: PMC3649863).
- Janecek JK, Swanson SJ, Sabsevitz DS, Hammeke TA, Raghavan M, Binder JR. Naming outcome prediction in patients with discordant Wada and fMRI language lateralization. Epilepsy and Behavior, 2013, 27: 399-403.
- Janecek JK, Winstanley FS, Sabsevitz DS, Raghavan M, Mueller W, Binder JR, Swanson SJ. Naming outcome after left or right temporal lobectomy in patients with bilateral language representation by Wada testing. Epilepsy and Behavior, 2013, 28: 95-98.
- Lee D, Swanson SJ, Sabsevitz DS, Hammeke TA, Winstanley FS, Possing ET, Binder JR. FMRI and Wada studies in patients with interhemispheric dissociation of language functions. Epilepsy and Behavior, 2008; 13: 350-356.
- Sabsevitz DS, Swanson SJ, Hammeke TA, Spanaki MV, Possing ET, Morris GL, Mueller WM, Binder JR. Use of preoperative functional neuroimaging to predict language deficits from epilepsy surgery. Neurology, 2003; 60: 1788-1792.
- Stewart CC, Swanson SJ, Sabsevitz DS, Rozman ME, Janecek JK, Binder JR. Predictors of language lateralization in temporal lobe epilepsy. Neuropsychologia, 2014; 60: 93-102.
- Umfleet LG, Janecek JK, Quasney E, Sabsevitz DS, Ryan JJ, Binder JR, Swanson SJ. Sensitivity and specificity of memory and naming tests for identifying left temporal-lobe epilepsy.
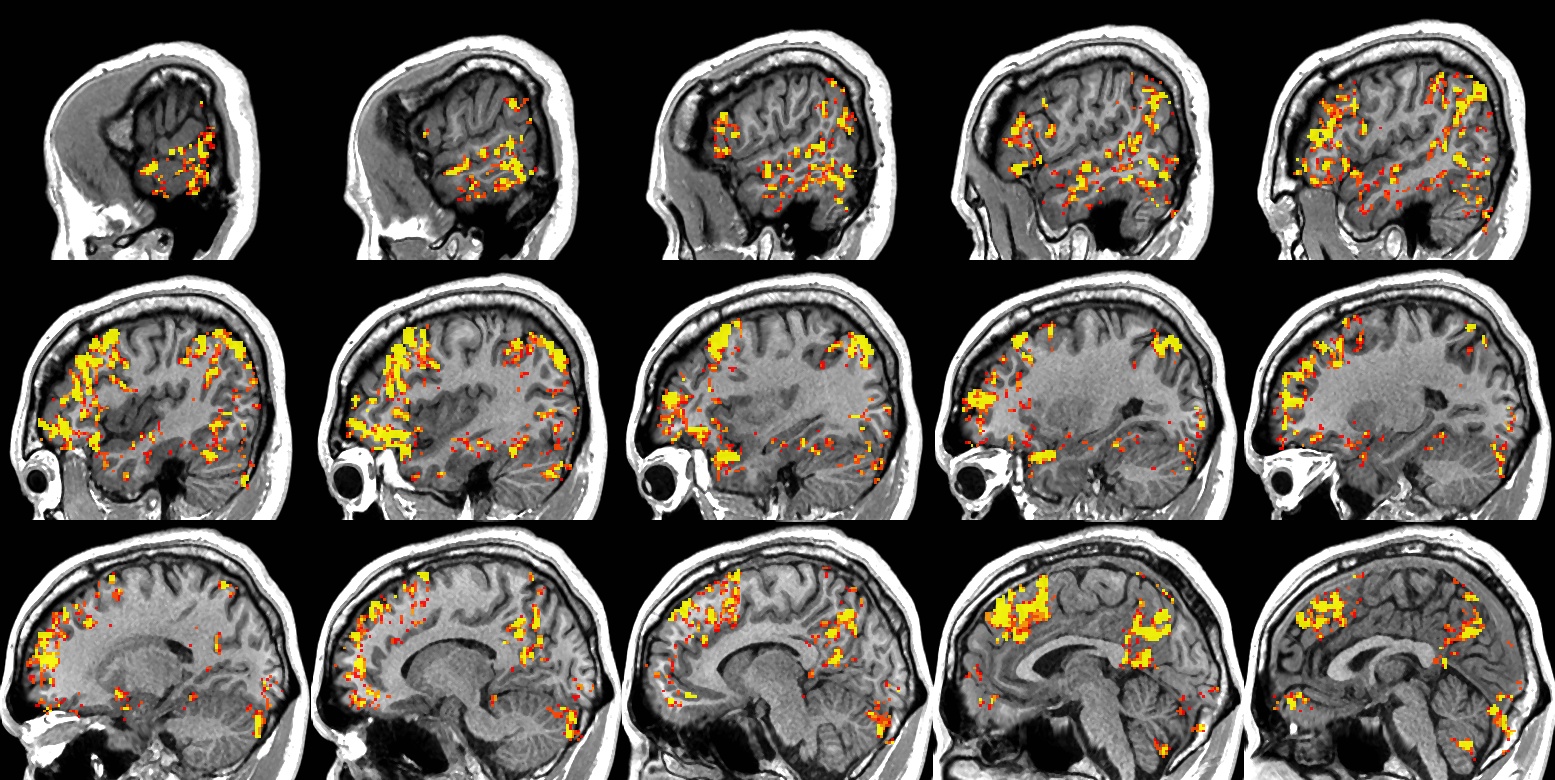
The Epilepsy Connectome Project
Funding source: NIDCD U01 NS093650 (Binder, Meyerand, Co-PIs)
Partial epilepsy is increasingly viewed as a network-level phenomenon involving progressive changes in brain morphology and inter-regional connectivity. Preliminary evidence suggests that these changes may be related to duration and severity of epilepsy, severity of cognitive and psychiatric co-morbidities, and variation in medication responsiveness. The Epilepsy Connectome Project (ECP) aims to test these hypotheses by acquiring detailed neuroimaging and clinical data in a large (n = 200), prospective sample of patients with temporal lobe epilepsy who vary in epilepsy duration, seizure burden, and neurobehavioral status. MRI acquisitions closely follow the Human Connectome Project guidelines and include high-resolution structural imaging, 40 minutes of “resting state” fMRI, 4 cognitive task activation protocols (emphasizing language, semantic memory, emotional face processing, and social cognition), and high-resolution diffusion tensor imaging. Magnetoencephalography (MEG) acquisitions include resting state and 3 task activation protocols. Neurobehavioral assessments cover a wide range of domains using instruments from the NIH Toolbox, standard clinical measures, and experimental measures.
Relevant Publications
- Bonilha L, Jensen JH, Baker N, Breedlove J, Nesland T, Tabesh A, Lin JJ, Drane DL, Binder JR, Kuzniecky RI. The brain connectome as a personalized biomarker of seizure outcome after temporal lobectomy. Neurology, 2015; 5; 84:1846-1853.
- Cook CJ, Hwang G, Mathis J, Nair VA, Allen L, Almane DN, Birn R, Conant LL, DeYoe EA, Felton E, Forseth C, Hermann B, Humphries CJ, Kraegel P, Maganti R, Nencka A, Nwoke O, Ustine C, Raghavan M, Rivera-Bonet C, Rozman M, Struck A, Tellapragada N, Ward BD, Prabhakaran V, Binder JR, Meyerand ME. Effective connectivity within the default mode network in left temporal lobe epilepsy: Findings from the Epilepsy Connectome Project. Brain Connectivity, in press.
